Periodic table of elements atomic weight1/15/2024  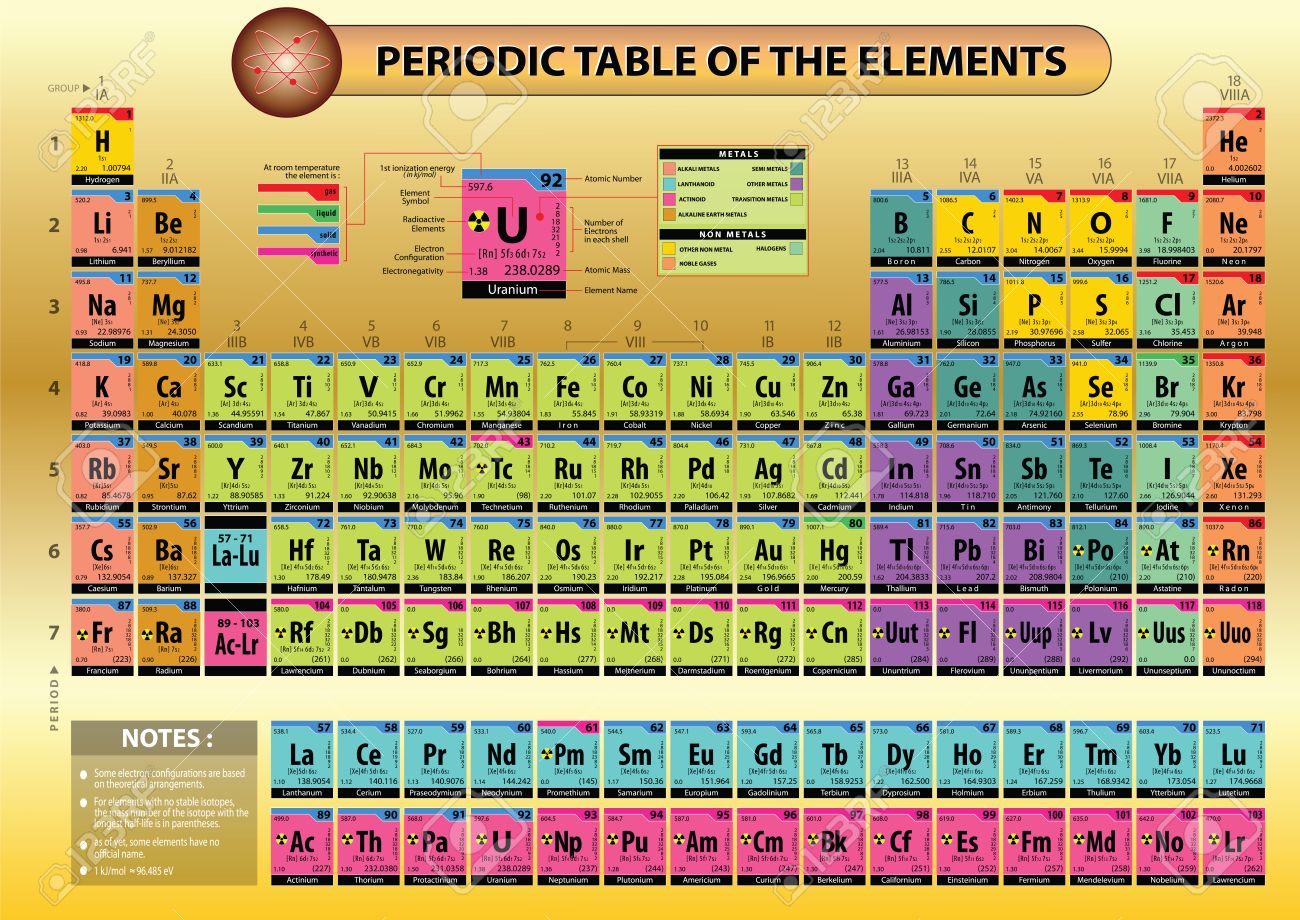
Elements in the periodic table are arranged according to the increasing atomic number, not the atomic weight, but often, atomic weight increase can also be seen between the consecutive elements when they are arranged according to their atomic numbers.The periodic table is an organised list of all chemical elements in order of increasing atomic number-that is, the total number of protons in the atomic nucleus.(This is because the mass of an electron is very small compared to a proton or a neutron). Number of protons and neutrons are largely contributing to the atomic weight.Atomic weight is the average weight calculated considering all the masses of isotopes. Atomic number of an element is the number of protons it has in the nucleus.What is the difference between Atomic Number and Atomic Weight? The weights given in the periodic table are calculated like this, and they are given as relative atomic mass. “An atomic weight (relative atomic mass) of an element from a specified source is the ratio of the average mass per atom of the element to 1/12 of the mass of an atom of 12C.” IUPAC defines the atomic weight as follows: For our purposes, we are using another unit call atomic mass unit (amu) to measure the atomic weight. 
Moreover, the masses of atoms are extremely small, so we cannot express them in normal mass units like grams or kilograms. 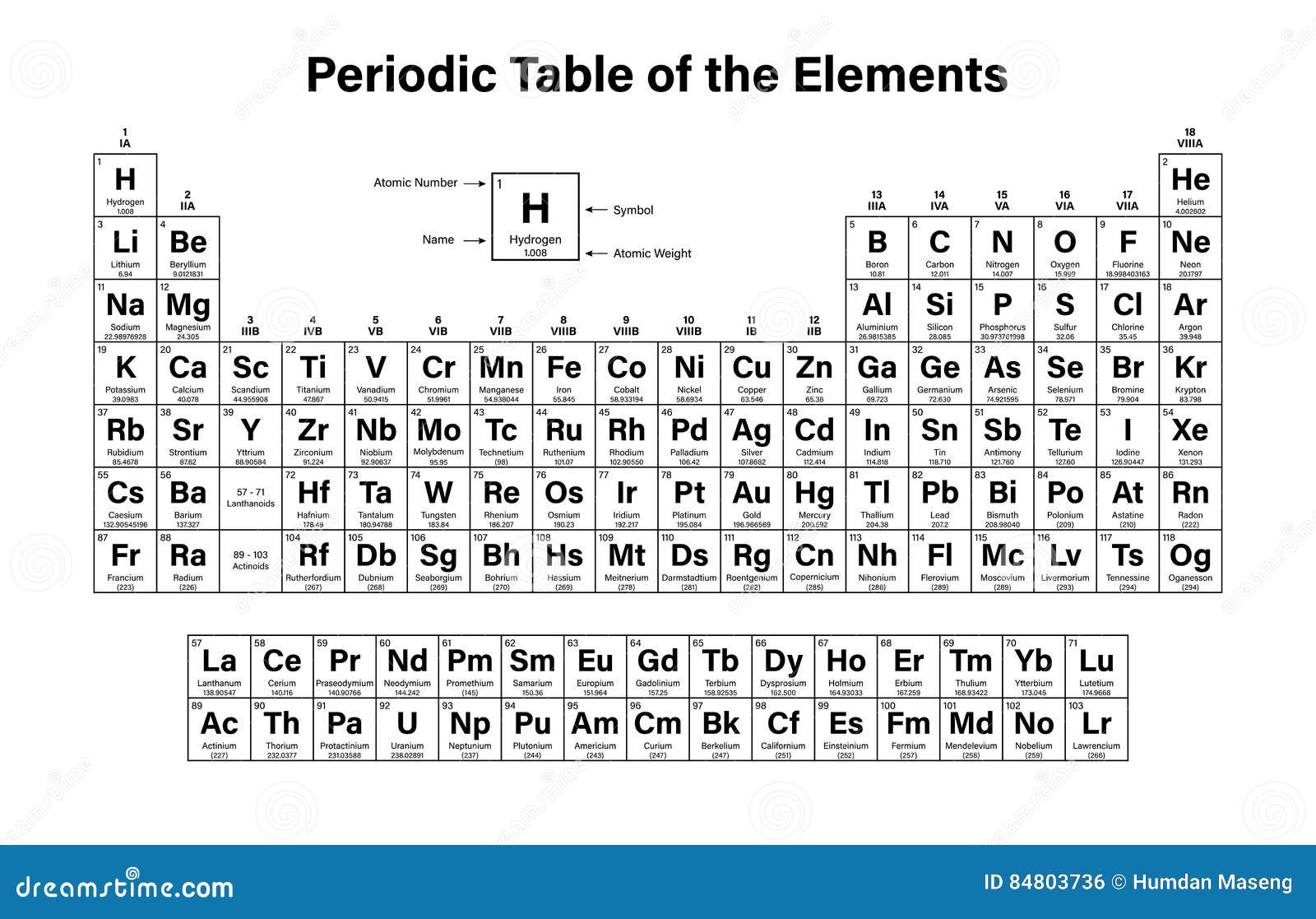
When calculating the atomic weight, both isotope mass and their relative abundances are taken into consideration. Each isotope is present in the environment, in different percentages. Since their neutron amount is different, each isotope has a different atomic mass. Isotopes differ from each other by having a different number of neutrons, even though they have the same proton and electron amount. Most of the atoms in the periodic table have two or more isotopes. Further, it gives information about oxidation states, charge of the ion, bonding behavior, nucleus charge, etc. For example, it tells the group and the period where the element belongs to in the periodic table. By looking at the atomic number itself, a lot of information about the element can be withdrawn. Therefore, atomic number is a good way of distinguishing different elements. Every element has separate atomic number, and no element has the same atomic number. This arrangement has automatically arranged them in increased atomic weight most of the time. The elements in the periodic table are arranged according to the increasing atomic number. However, it is always reliable to get the number of protons as the atomic number. Thus, atomic number is equal to the number of electrons in this instance. When the atom is neutral, it has the same number of electrons as protons. 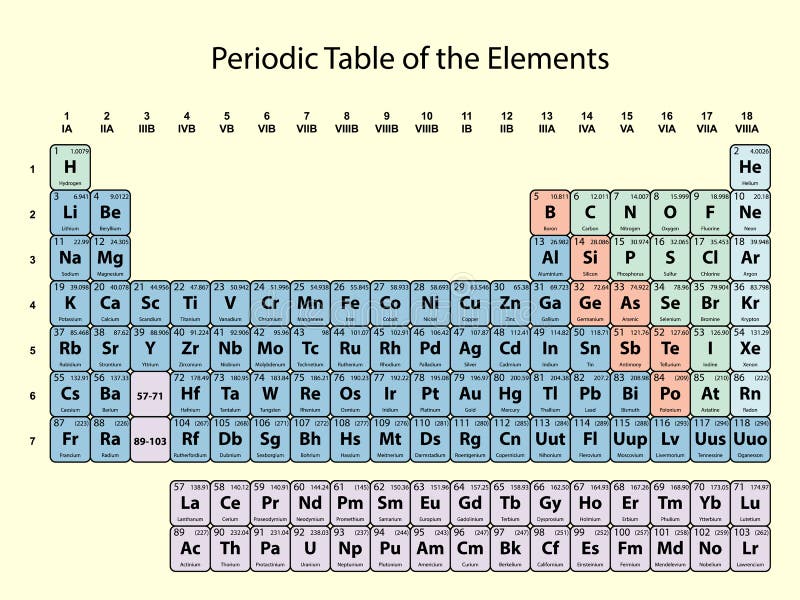
The symbol for denoting the atomic number is Z. Atomic number of an element is the number of protons it has in the nucleus. In addition, there are electrons circling around the nucleus in orbitals. Nucleus of the atom contains protons and neutrons. Atomic weight is also important to get some idea about the elements.Ītoms are mainly composed of protons, neutrons and electrons. Atomic number provides a variety of information regarding the atom and its nature. In the periodic table, atoms are arranged according to their atomic number. Atoms are characterized by their atomic numbers. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
